Amniotic Fluid Embolism Facts
A Clinician’s Guide to AFE
What is Amniotic Fluid Embolism (AFE)
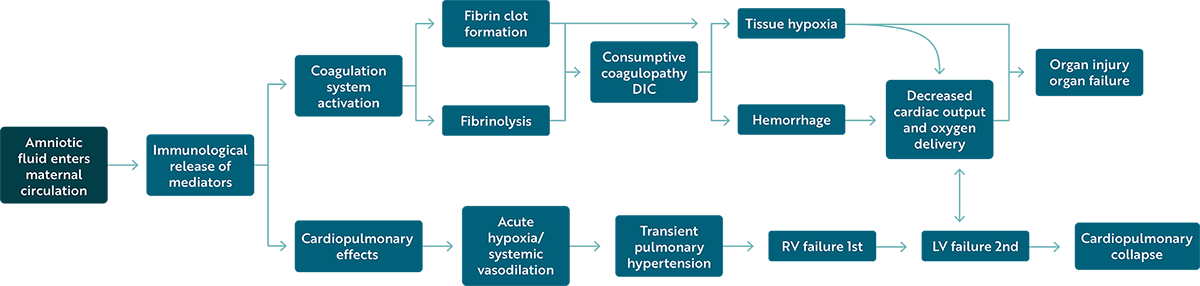
Amniotic fluid embolism (AFE) is characterized by a sudden and unexpected cardio-respiratory collapse and disseminated intravascular coagulopathy during labor or at the time of birth. Presentation of symptoms includes a triad of acute hypoxia, hypotension, and bleeding. It is thought to be a result of an anaphylactic-like reaction to amniotic fluid in the maternal circulation, a normally benign result of the birthing process. Although rare, it is often fatal and remains a leading cause of maternal morbidity and mortality in the United States. It may also be referred to as anaphylactoid syndrome of pregnancy.
Key Points
- Suspect AFE with classic symptom triad: Hypoxia, Hypotension, Coagulopathy
- Treatment is aggressive and supportive.
- AFE is a clinical diagnosis of exclusion – fetal cells in the vasculature are not confirmative nor diagnostic.
Incidence
The true incidence of AFE is unknown but is estimated at 1 in 40,000 births, with a mortality rate ranging from 20-60%. Rates will vary depending on where data is derived. For large administrative data sets, the incidence rates increase and severity decreases. This is likely due to over reporting and data selection criteria including all ICD-10 codes or including forme fruste cases that do not meet the classic definition. Data from case reports, such as those in the AFE Registry, are more likely to have a lower incidence and a higher rate of severity as cases have been reviewed by AFE experts.

A leading cause of maternal morbidity and mortality

20-60%
Mortality Rate

1 in 40,000
deliveries
Risk Factors
AFE remains an unpredictable and therefore unpreventable event. Data regarding risk factors for AFE are inconsistent and contradictory. As a result, changes to obstetric practice to reduce the risk of AFE are not recommended.
Pathophysiology
AFE appears to result from an abnormal maternal response to amniotic fluid entering the mother’s bloodstream during labor and birth. Entry of fetal cells and amniotic fluid into the bloodstream is a normal occurrence. However, some mothers mount a massive, abnormal immune response similar to anaphylaxis. Pulmonary obstruction is no longer considered a pathophysiologic component of AFE.
Clinical Presentation and Diagnosis
AFE is a clinical diagnosis. The diagnostic criteria for AFE for research related includes:
- Sudden onset of cardiopulmonary arrest, or both hypotension with respiratory compromise
- Documentation of overt DIC following the appearance of initial signs and symptoms
- Clinical onset during labor or within 30 minutes of placenta delivery
- No fever during labor
Early Signs of Symptoms
- Anxiety
- Agitation
- Impending sense of doom
- Confusion
- Shortness of breath
- Nausea and vomiting
- Fetal bradycardia
In atypical cases, one or more of these signs may be absent. Careful exclusion of other conditions are essential. The identification of fetal squamous cells in the maternal circulation occurs routinely and is not diagnostic of AFE. Diagnostic criteria also differs among counties. The U.S. holds the most stringent diagnostic criteria helping to exclude other more common causes of severe maternal complications such as postpartum hemorrhage, hemorrhagic shock, pulmonary embolism, sepsis and septic shock, anaphylaxis, and medication reactions.
Management
Treatment of AFE is aggressive and supportive. If the patient is in cardiac arrest, the first step is to provide high quality cardiopulmonary resuscitation and initiate Advanced Cardiac Life Support (ACLS). If undelivered, manual left uterine displacement is recommended to relieve aortal-caval compression with plans to proceed with resuscitative cesarean section after 4-5 minutes if spontaneous circulation has not returned. Respiratory support should be provided with anticipation of need for intubation and mechanical ventilation. Coagulopathy occurs even without immediate evidence of hemorrhage and requires anticipation with aggressive and rapid correction with blood products. Learn more.
Anecdotal Therapies
There have been significant discussions and concerning assumptions about the use of Atropine, Ondansetron, and Ketorolac ( “A-OK” ) as an effective and novel approach to treat AFE. The literature around the use of these medications is especially limited and based on a small number of case reports. Of those case reports, the authors acknowledge the case does not meet diagnostic criteria. There is a confirmation bias in the literature on the use of A-OK in AFE, as cases with poor outcomes associated with A-OK use do exist but are not reported.
Focus should be on performing high-quality ACLS with cardiovascular support, transfusion therapy, and resuscitative delivery, along with the use of approved pharmacologic agents. If these interventions do not result in the return of spontaneous circulation, use of other therapeutic agents can be considered as a management strategy used by the primary care team caring for the patient. However, they should never be prioritized over standard ACLS interventions. Clinicians are encouraged to practice ACLS through demonstration of skills and participation in regular maternal arrest simulations to prepare for this rare but highly acute event.
Recurrence of AFE in Subsequent Pregnancies
Recurrence risk of AFE is not known due to the rarity of the condition, limited data, and many inaccurate diagnoses. However, cases of successful pregnancies following AFE have been reported to the AFE Foundation. It is believed that the risk of recurrence is the same as the risk of initial incidence. The AFE Foundation offers supportive services to patients and clinicians treating patients who wish to have a subsequent pregnancy following an AFE. Learn more.
Current Research
Due to its unpredictable nature and high mortality, AFE is exceptionally difficult to study. Many animal models that were developed in the years following the initial publications by Steiner and Lushbaugh in 1941 were unsuccessful in replicating AFE as they were largely based on the theory that AFE was a mechanism of pulmonary obstruction. Ultimately, what they did prove was that entrance of amniotic fluid into the central circulation was largely inconsequential.
In 2012, the AFE Foundation along with a team of researchers at Baylor College of Medicine and the Perinatology Branch of the National Institutes of Health established the AFE Registry, an international database of self-reported cases. In 2022, the AFE Registry was moved to the University of Texas Health Science Center at Houston and expanded to include a tissue and specimen biorepository. This database of cases, the largest ever amassed, allows for the examination of over 650 data points related to treatment, management and demographic data. While this data is important and continues to be collected, it has not aided in uncovering the true etiology of the condition nor has it allowed us to identify why some patients respond to treatments while others do not.
Our AFE Registry has allowed us to better understand the clinical course of AFE and examine over 650 data points related to treatment, management and demographic data. While this data is important, it has not aided in uncovering the true etiology of the condition nor identify why some patients respond to treatments while others do not.
Clinical Trials: NCT05791786